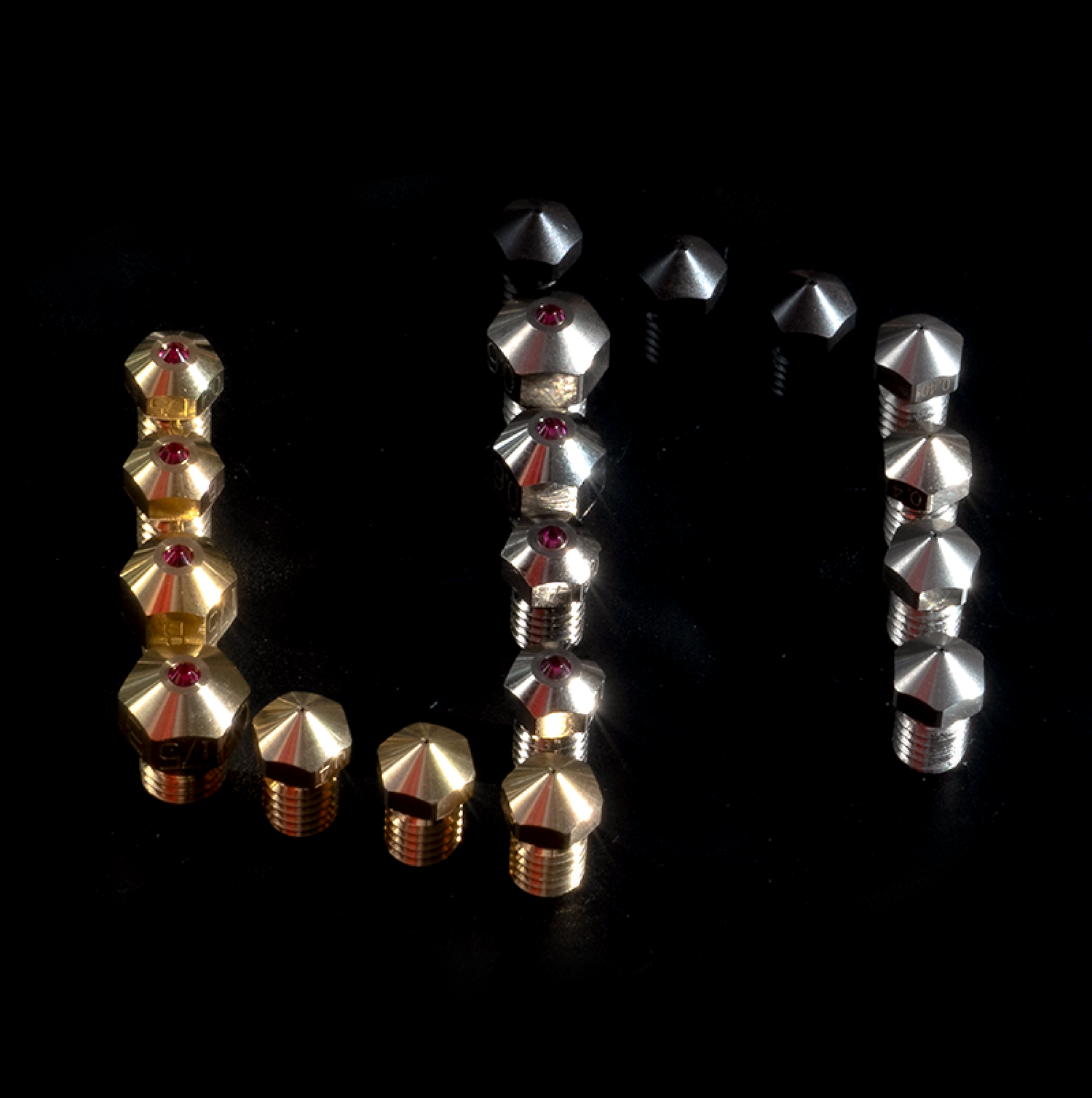3D打印机核心零部件供应商
公司简介
锐力斯是一家专注于3D打印机核心零部件研发、生产、销售的国家高新技术企业。面向国内外3D打印机市场提供热端、挤出机、传动系统核心组件的OEM/ODM定制化解决方案。自2013年创立以来,锐力斯始终秉承科技、创新、标准、服务的发展理念......
产品中心
锐力斯定位高端3D打印上游市场,专注研发、生产3D打印机核心零部件产品,现拥有热端、挤出机、皮带&带轮、直线轴承、电机等五大系列主流产品,为相关客户提供喷嘴、喉管、散热体、加热块、挤出轮、皮带、带轮、直线轴承及电机等上百种3D打印机核心零部件产品。
核心优势
多年来,锐力斯始终坚持品质至上、诚信为本、互利共赢的发展理念,在实践中不断积累经验、改进不足,完善流程,最终探索出了一套专业高效的生产管理&客户服务体系……
-

强大的加工能力
锐力斯先进的生产设备和检验仪器、专业可靠的技术团队,科学高效的管理体系、ODM/OEM个性化定制
-

可靠的质量保证
来料检验,生产自检(包括首件检验),过程巡检、外观全检以及出货检验,五大质检全方位预防并把控产品质量
-

完善的客户服务
专业的技术团队,将为客户提供不同环节的产品咨询服务,解决相关技术问题!完善的客户服务使您免于后顾之忧
合作伙伴
凭借卓越的产品质量、强大的生产实力和完善的售前售后服务,锐力斯被越来越多的客户认可,与国内外诸多知名3D打印厂商形成稳定可靠的战略合作关系,自成立以来,服务客户超过3000+,产品已经销往世界各地共90多个国家和地区
邮件订阅
成为第一个知道锐力斯新品系列的人